We are the NIHR Clinical Research Network:West Midlands – Young Research Champions Group (YRC).
We were established in September 2009 and are proud to be part of the Generation R initiative.
Over many years we have worked with many Chief Investigators, Research and Development Departments and Commercial Partners to help with the design, development and delivery of research.
We meet once a month in Birmingham. Meetings take place on Saturdays 10.30am-3.30pm
We are a diverse group of about 20 young people, between the ages of 9 and 18 years of age . Some members may be at full time school, college, working or even studying at University. We have all accessed healthcare and some of us have mild or chronic healthcare conditions or are carers. These conditions may affect both physical and/or mental health.
If you would like to find out how you could become a member then email carly.tibbins@nihr.ac.uk
We are currently recruiting new members and want to hear from you!
We live and experience what it’s like to be young people. Our views are crucial to ensure that research for children and young people is the best it can be.
You can keep up to date with our activities by following us on Twitter @YPSGWMids
You can watch some of our own films that give examples of the type of activities we are involved in. These activities include:
Helping Chief Investigators with the design, delivery and dissemination of research studies https://m.youtube.com/watch?v=JGV0gnFteDQ
Helping to develop training materials for other children and young people. We have even attended and presented our activities at Patient and Public Involvement and Engagement training sessions at hospitals https://m.youtube.com/watch?v=8MuUvD0hbs0
We raise awareness about research
https://m.youtube.com/watch?v=lS2VfzbVYZ4
We work with Research and Development departments at NHS Trusts as Patient Research Ambassadors https://m.youtube.com/watch?v=HZb26M1uMpM
Scroll down this page to find out what we have been up to recently.
Testimonials
The NIHR CRN West Midlands Young Research Champions Group (YRC) recently received the following feedback.
Industry Partner
“I used the review service offered by your YPSG to review Patient Information Sheet-Informed Consent Forms for a paediatric study. I was very impressed with the input I received; it was well detailed and for all changes suggested, rationale was provided. The quality of the feedback was excellent and I was able to implement the changes suggested and submit to the Research Ethics Committee (REC) in a timely manner. The REC feedback was very positive and approval was received very quickly. The responsiveness and professional conduct of the NIHR CRN team is excellent. This is a wonderful service that I wish I had used in the
past it would have saved me a lot of additional work.
Nicki Vanes, Clinical Manager and Lead Nurse – NIHR/Wellcome Trust Clinical Research Facility (Birmingham Children’s Hospital)
“The enthusiasm form the NIHR CRN West Midland’s YPSG members was electric, they were so committed to getting the filming just right, to capture the patients journey through the Clinical Research Facility here at BCH. We aim to make this video part of a suite of new IT APPs/files which can be accessed by our patients and their families before they start on a research study here at BCH CRF, making a visit to us as good as possible, assist with true informed consent and assist the team here to develop real individualised patients pathways and care. We are so grateful for YPSG members in assisting with this development.”
Our current members joined the group by a variety of different routes including referrals from their doctors from different West Midlands hospitals, schools, word of mouth or work experience placements.
We meet up once a month at a city centre location in Birmingham. Meetings take place on Saturdays from 10.30am until 3.30pm.
Members of our group are also involved in representing the NIHR Clinical Research Network: West Midlands by attending National and International conferences or meetings, being a member of another group or committee and presenting the groups activities at conferences and meetings.
Link To iCAN: International Children’s Advisory Network Summit -Barcelona June 2016
So what do the YPSG members say about their involvement
“I’ve learnt lots about the different stages if a clinical trial and what’s involved.”
“I’ve learnt how long it takes for a medicine or new drug to be developed, different phases the medicines goes through and the time it takes to create one.”
“I’ve made some great friends and being in the group has really helped with my personal confidence.”
“Being a member of the group has given me lots of opportunities and helped me develop some great skills!”
” It’s great to know that we are helping other children and young people and improving research!”
Contact
We are here to help! Are you a Chief Investigator, Research and Development Department or Research professional? We are waiting to hear from you!
The West Midlands group is facilitated Carly Tibbins , Patient and Public Involvement and Engagement Officers, NIHR Clinical Research Network: West Midlands
Email: Carly.tibbins@nihr.ac.uk

Working together teaching postgraduate students how to carry out research with young people since February 2020! Read on to for a combined Blog from three of this year’s GOSH YPAG member attendees as they reflect on their annual visit in February 2026 to work with over 30 students Thank you to Aleena Lu’Lu, Betty and […]

A big thank you to GOSH YPAG member Teo who hosted @ GreatOrmondSt Twitter account, to give visitors the inside scoop of the day’s activites at this year’s NIHR GOSH BRC Family Fun Day for research on 26 October 2019. Teo is a dedicated YPAG member and has been involved in lots of other activities to represent […]
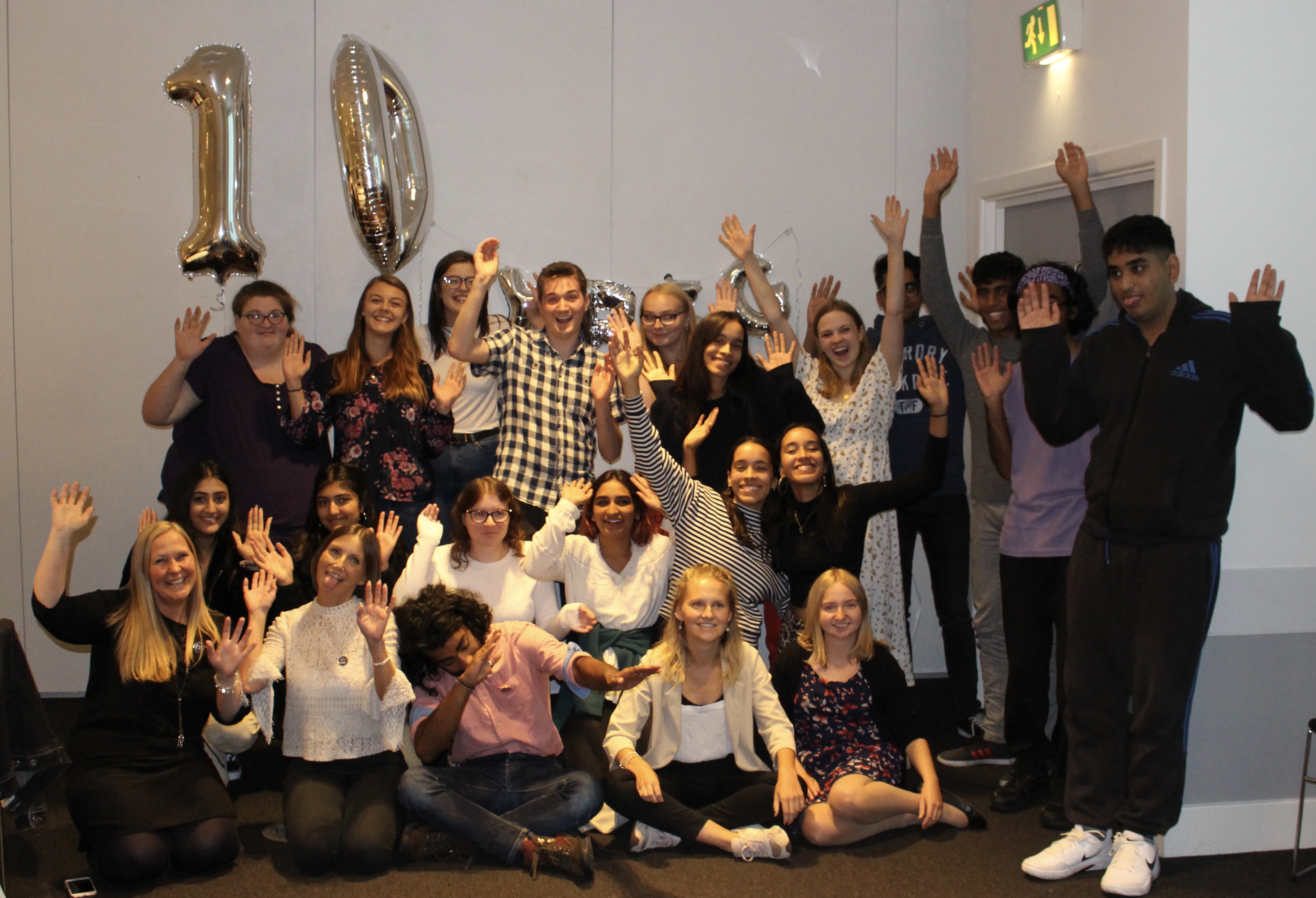
The West Midlands Young Persons’ Steering Group had a 10 year celebration last month. It was a great day where current and past members attended to talk about their experience of the group and how it’s impacted on their education and career. The day was made up of a mixture of video’s and presentations from […]